
Dr Vilaiwan (Vil) Fernandes is a developmental neurobiologist at UCL. Her lab studies the Drosophila visual system to uncover fundamental principles of brain development. She speaks passionately about the importance of mentors in her career so far and why the fly could help crack the mysteries of neuronal reprogramming.
We recently caught up with Vil to find out more about her research into the neurobiology of the fruit fly.
Q: Your path into developmental neurobiology wasn’t straightforward. How did it unfold, and who shaped it?
A: I actually studied marine biology, ecology and evolution, and mathematics as an undergraduate – not cell or developmental biology. Back then, I thought cell and developmental biology was quite boring. The turning point was an immersive course at Friday Harbor Labs that I applied to because I mistakenly assumed it would be about ecological networks. When I arrived, I realised it was actually about computational modelling of gene regulatory networks in cells. But it was absolutely thrilling. It showed me how little we know and how many open questions there are in cell and developmental biology, as opposed to the “everything is solved” feeling you sometimes get from undergraduate textbooks.
At the end of the course, I stayed on as a research assistant and worked on modelling robustness in the Drosophila segment polarity network. That experience made me want to learn the experimental side too, so I applied for PhDs in cell and developmental biology and eventually ended up – somewhat by chance – in a Drosophila lab. When choosing a postdoc, the main thing I wanted was a lab with a good atmosphere. In the end, the Drosophila lab that I had imagined would just be “practice” for my interviews turned out to be the best fit.
Q. So, we have to say it. WTF?!
A: Haha, you were listening to my talk at the annual meeting! “WTF – Why The Fly?” was a joke I have to admit stealing from Professor Jordan Raff at Oxford. But it does emphasise that Drosophila is great for answering neurodevelopmental questions.
The broad question for my group is simple to say but vast in scope: how are brains built? We’re interested in how neurons and glia are made from precursors, how they diversify, how the right numbers of each cell type are produced, and how timing is used as developmental information. Time keeps coming up again and again in development, and I find that very compelling.

We use the Drosophila visual system – especially the lamina – because it gives us exquisite experimental access. It’s a beautiful system. The lamina is patterned with single-cell precision: for every unit eye, there is a lamina column with six precursors that generate five neuron types. In previous work, my lab showed that the morphogen Hedgehog produced by photoreceptors forms a gradient along these columns which helps specify different fates.
But Hedgehog alone wasn’t enough to explain everything. We’ve now shown that glial populations infiltrate the lamina from the top and also send signals from the bottom. Crucially, the rate at which they arrive matters. If glia move in too quickly, the timing of Delta-Notch signalling is thrown off. The integration of Hedgehog, timing cues from glia, and other pathways ultimately produce the precise pattern.
The broad question for my group is simple to say but vast in scope: how are brains built?
Q: Your lab recently challenged assumptions about glial diversity. What did you discover?
A: There’s been a long-running debate because astrocytes and other glia have striking morphological diversity. Astrocytes show very different shapes across and within brain regions; does different morphology mean different cell types, as we often assume for neurons? Or are these cells morphologically plastic, as people have observed especially after injury. What exactly is the link between shape, function and cell type?
During the pandemic, when lab work was limited, we analysed single-cell transcriptomic datasets from the optic lobe, and built an atlas of glial morphologies from the literature and our imaging. We did find some genuine transcriptional diversity, but morphology was a poor predictor of it. Often, multiple morphologies mapped to the same transcriptomic cluster. Our interpretation is that the same cell type adopts different shapes depending on the local environment – like a multilingual person who speaks a different language depending on who they’re talking to.
That runs counter to what a lot of people expected, so it’s controversial. A priority now is to pin down the specific environmental interactions that drive those shapes and to understand the impact that different morphologies have for these cells and the neurons they take care of, so that we can properly reconcile the field.
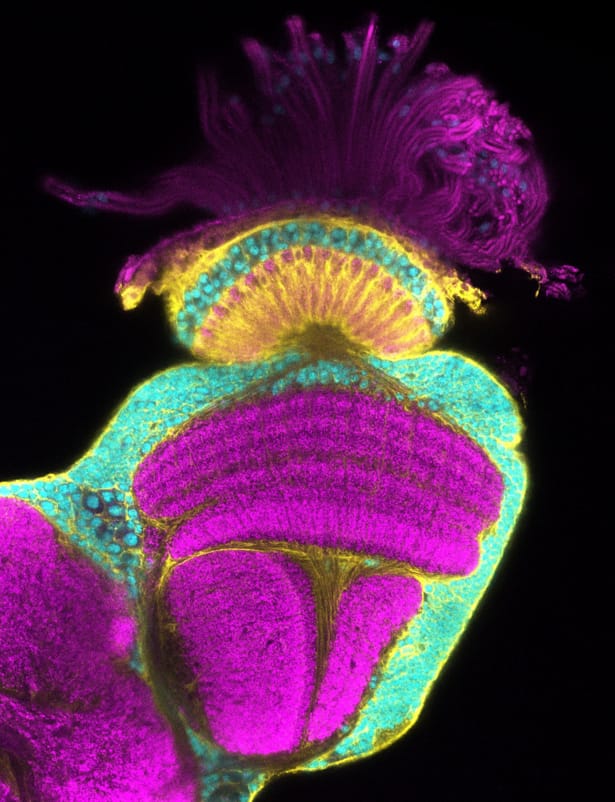
This confocal micrograph of an adult Drosophila melanogaster optic lobe shows the remnants of a compound at the very top. Neuronal cell bodies (cyan) surround the synapse dense neuropils (magenta). In yellow are glial membranes, which infiltrate every part of the tissue.
Q: You’ve spoken about a “general neuronal programme”. What does that mean for understanding how neurons are made?
A: All neurons share certain core traits – being post-mitotic, extending axons and dendrites, forming synapses – yet we know surprisingly little about how this shared programme is triggered compared with how specific neuron subtypes are specified. This gap matters not only for basic neurodevelopment but also for reprogramming and regeneration.
Our research is showing that there is a small early network of factors that cells must switch on to begin the neuronal programme and a set of precursor programmes they must switch off. Both sides of that equation matter. I had perhaps been naïve in thinking mostly about the genes that need to be turned on; but you also need to actively suppress features of the previous state.
We also see post-transcriptional brakes in precursors – mechanisms that keep neuronal proteins from being made prematurely. During differentiation, those brakes are released at the same time as the core neuronal programme is activated. That combination helps tip a precursor cell decisively into a neuronal state.
The idea of direct neuronal reprogramming is exciting because it’s faster than going through an induced pluripotent stem cell state, avoids some risks, and can preserve aspects of cellular age. But efficiency is low, and cells often get stuck part-way. I think that many of those bottlenecks come from failures in general neuronal differentiation, not necessarily neuron subtype identity.
Our work gives us a much more rational starting point. In flies, mis-expressing a single transcription factor can push non-neuronal cells part-way towards neurons in vivo, and the hurdles resemble those seen in mammalian reprogramming: low viability, low efficiency, instability. That’s helpful because it means we can use the fly as a platform to test combinations – promoting the early neuronal programme while relieving the precursor‑state barriers – in a systematic way.
Many of the factors we’re identifying are evolutionarily conserved, so we’re optimistic that the logic will translate.
Q: What has receiving the Lister Prize meant for you and your team?
A: I’m genuinely honoured to be part of the Lister community – and yes, still a bit intimidated. The Annual Meeting was wonderful; people were very engaged, and I felt comfortable asking questions across disciplines. That really reminded me of the generosity of people’s time and attention I first experienced at Friday Harbor.
The funding gives me breathing room. It lets me be more ambitious, attempt experiments that take longer, and support postdocs so they can produce their best work without a looming deadline. The project on the general neuronal programme actually began as a PhD student’s “side hustle”, and it wasn’t covered by other grants, so the Lister Prize has allowed us to develop it properly.
Being part of the Lister community is opening doors. It reminds me of the importance of mentorship in research. Garry Odell at Friday Harbor was a polymath – an encyclopaedia of knowledge – who could dismantle a confocal microscope with you in the afternoon and turn it into a lesson. That research centre was very close‑knit, and Garry created an environment where you were never intimidated to ask questions. I still look for that feeling and it’s something I recognise in the Lister community today: a place where you can ask questions freely without feeling foolish.

The funding gives me breathing room. It lets me be more ambitious, attempt experiments that take longer, and support postdocs so they can produce their best work without a looming deadline.
Find out more
- Fernandes Lab (UCL website)
- Fernandes Lab (Lab website)



